MANGANESE(II) HYDROGEN PHOSPHITE MONOHYDRATE
Manganese(II) hypophosphite monohydrate
CAS: 7783-16-6
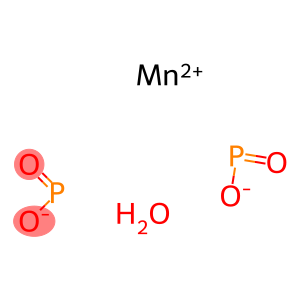
Molecular Formula: H2MnO5P2
MANGANESE(II) HYDROGEN PHOSPHITE MONOHYDRATE - Names and Identifiers
| Name | Manganese(II) hypophosphite monohydrate |
| Synonyms | MANGANOUS HYPOPHOSPHITE Manganesehypophosphitemonohydrate Manganese Hypophosphite Monohydrate Manganesehypophosphite monohydrate Maganese(II)hypophosphite monohydrate MANGANESE(II) HYPOPHOSPHITE-1-HYDRATE MANGANESE(II) HYPOPHOSPHITE-1- HYDRA MANGANESE(II) HYPOPHOSPHITE MONOHYDRATE Manganese(II) hypophosphite monohydrate MANGANESE(II) HYDROGEN PHOSPHITE MONOHYDRATE Phosphinic acid, manganese(2+) salt, monohydrate phosphinolate, oxo-, manganese(2+) salt, monohydrate |
| CAS | 7783-16-6 |
| EINECS | 677-616-7 |
| InChI | InChI=1/Mn.HO2P.H2O/c;1-3-2;/h;(H,1,2);1H2/q+2;;/p-1 |
| InChIKey | DIJYHROXKPCRKH-UHFFFAOYSA-L |
MANGANESE(II) HYDROGEN PHOSPHITE MONOHYDRATE - Physico-chemical Properties
| Molecular Formula | H2MnO5P2 |
| Molar Mass | 198.9 |
| PH | 3.5-4.5 (20℃, 5%) |
| Storage Condition | Room Temprature |
| MDL | MFCD00150256 |
| Physical and Chemical Properties | This product is white or flesh white crystals. Soluble in No, easy hydrolysis, strong hygroscopicity. Insoluble in alcohol. |
MANGANESE(II) HYDROGEN PHOSPHITE MONOHYDRATE - Risk and Safety
| Risk Codes | R20/21/22 - Harmful by inhalation, in contact with skin and if swallowed. R36/37/38 - Irritating to eyes, respiratory system and skin. |
| Safety Description | S26 - In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. S36 - Wear suitable protective clothing. |
| WGK Germany | 3 |
MANGANESE(II) HYDROGEN PHOSPHITE MONOHYDRATE - Reference Information
| EPA chemical substance information | information provided by: ofmpeb.epa.gov (external link) |
| Use | This product is used as a ferrous metal corrosion inhibitor in the electroplating industry, and is also widely used for phosphating and rust prevention of steel products. |
| production method | put 110 of manganese sulfate into the dissolution tank, add water to dissolve, and then drive into the primary converter. Then, 170 of soda ash was added to the dissolution tank, and water was added to dissolve the solution. After the solution was added to the primary conversion kettle under stirring, the reaction was completed when the pH value reached 7.5 to 8.0. After being allowed to stand, the upper layer was removed by suction, and the lower layer was precipitated with manganese carbonate. After washing with distilled water, phosphoric acid was added dropwise to pH 4 to obtain trimanganese phosphate, and the trimanganese phosphate was washed with distilled water. Then transfer the material to the secondary converter, continue to add 170kg 85% Phosphoric acid, when the concentration of the solution to 30~35 ° Bé, add the appropriate amount of barium carbonate, after stirring at 70-80 °c for 0.5h and standing for 24 h, the barium sulfate precipitate was removed, and the filtrate was transferred to a vacuum evaporator and concentrated until crystals precipitated. Centrifugal dehydration, crystallization and drying to obtain the finished product. Reaction formula: |
Last Update:2024-04-10 22:29:15
Supplier List
Spot supply
Product Name: Manganese hypophosphite monohydrate Visit Supplier Webpage Request for quotationCAS: 7783-16-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Manganese(II) hypophosphite monohydrate Request for quotationCAS: 7783-16-6
Tel: 86+027-83389957
Email: 1024042217@qq.com
Mobile: 86+18627766980
QQ: 1024042217
Wechat: 18627766980
Product Name: MANGANESE(II) HYPOPHOSPHITE MONOHYDRATE Request for quotation
CAS: 7783-16-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7783-16-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Manganese(II) Hypophosphite Monohydrate Visit Supplier Webpage Request for quotation
CAS: 7783-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7783-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: Manganese hypophosphite monohydrate Visit Supplier Webpage Request for quotationCAS: 7783-16-6
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Manganese(II) hypophosphite monohydrate Request for quotationCAS: 7783-16-6
Tel: 86+027-83389957
Email: 1024042217@qq.com
Mobile: 86+18627766980
QQ: 1024042217
Wechat: 18627766980
Product Name: MANGANESE(II) HYPOPHOSPHITE MONOHYDRATE Request for quotation
CAS: 7783-16-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
CAS: 7783-16-6
Tel: +86 13454675544
Email: rachel@api-made.com
QQ: 3510434126
WhatsApp: +86 13454675544
Product Name: Manganese(II) Hypophosphite Monohydrate Visit Supplier Webpage Request for quotation
CAS: 7783-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 7783-16-6
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



